DM1
One repeat expansion. A cascade of splicing chaos. Myotonic Dystrophy type 1 is the most common adult-onset muscular dystrophy, where toxic RNA hijacks splicing regulators and progressively dismantles muscle function.
CYTOO’s MyoScreen™ platform recapitulates DM1 pathology in micropatterned primary human myotubes, enabling high-content detection of DMPK RNA foci, MBNL1 sequestration, splicing defects and downstream contractile dysfunction, giving your program the cellular-level evidence it needs to advance with confidence.
CYTOO's approach
At CYTOO, our MyoScreen™ platform utilizes physiologically relevant micropatterned muscle cultures derived from human primary donor cells to model molecular and functional features of DM1.
This system enables high-content imaging and AI-based analysis to sensitively capture hallmark DM1 phenotypes, including impaired myotube maturation, DMPK RNA foci in nuclei, dysregulated protein localization, and defects in calcium handling and contractile function.
These readouts support the evaluation of therapeutic strategies aimed at reducing toxic RNA, restoring normal splicing patterns, and correcting downstream functional consequences. Overall, CYTOO’s technologies provide an end-to-end platform to support DM1 drug development.
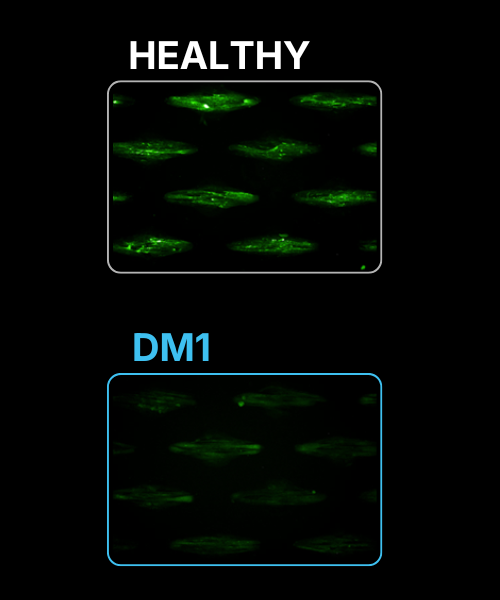
Adult specific dystrophin staining : Alteration of dystrophin splicing is present in DM1 donors as evidenced by lack of adult dystrophin signal
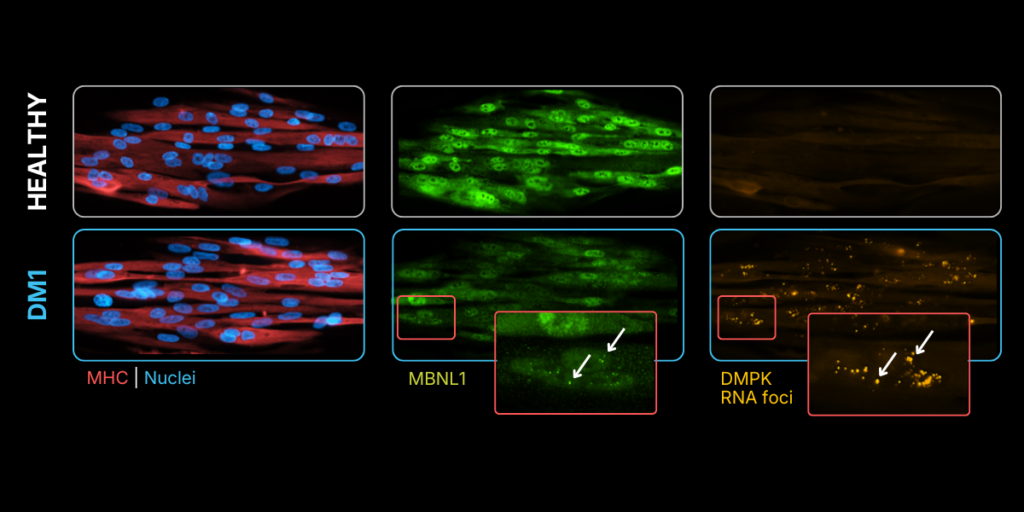
Detection of DMPK RNA foci and decrease of MBNL1 intensity in DM1 cells
About DM1
Myotonic dystrophy type 1 (DM1) is an autosomal dominant multisystem disorder caused by an expanded CTG trinucleotide repeat in the DMPK gene. This expansion leads to the accumulation of toxic RNA transcripts that sequester RNA-binding proteins, notably members of the MBNL family, resulting in widespread misplicing and reversion to an embryonic splicing program. In skeletal muscle, these splicing defects disrupt key pathways involved in excitation–contraction coupling, ion channel function, calcium homeostasis, and muscle differentiation, ultimately leading to muscle weakness, myotonia, and progressive atrophy. DM1 also affects cardiac conduction, respiratory function, and metabolic regulation, contributing to significant morbidity and reduced life expectancy. To date, no curative therapies are available, and disease-modifying treatments remain an important unmet medical need.
Explore our catalog of characterized donors and readouts
How can we work together
Everything is tailored to your needs through a flexible R&D partnership model that fosters true collaboration and innovation. We offer adaptable project structures and FTE allocation to fit your goals.
